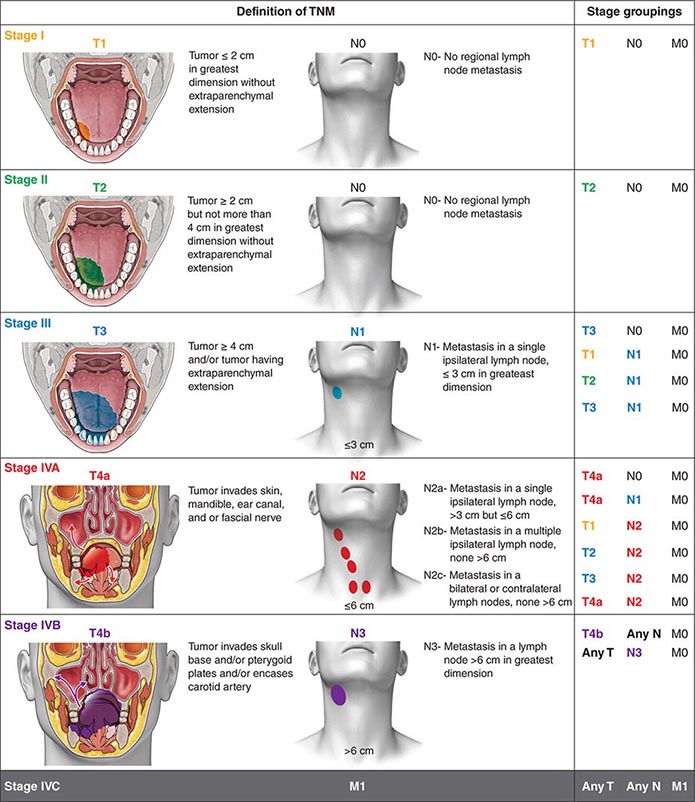
It must also be noted that the bifurcated treatment path described above was not created by CEL-SCI for its study but was, and still is, the SOC per NCCN Guidelines for advanced primary SCCHN patients. Note that the term “lower risk for recurrence” should not be confused with a low risk of death, because the 5-year survival for such patients is still less than 50%, even after receiving the current SOC. Patients would not meet the eligibility criteria if they present with adverse features that would place them in the higher-risk-for-recurrence group per NCCN Guidelines. The “Proposed Indication” for Multikine will be limited by certain eligibility criteria to select patients deemed by NCCN Guidelines for the lower-risk-for-recurrence group. All other patients were classified as lower risk for recurrence and would receive only radiotherapy after surgery. These patients would receive concurrent chemoradiotherapy. To determine which path a patient should take, the treating physicians decided after surgery based on pathology and medical judgment, as guided by the National Comprehensive Cancer Network (NCCN) Guidelines, whether the patient was at a higher risk for tumor recurrence. The SOC for all research subjects was identical and involved a bifurcated treatment path following surgery, i.e., a radiotherapy-only arm and a concurrent chemoradiotherapy arm. The two main comparator groups in the study were the primary arm (Multikine+CIZ+SOC) and the study control arm (SOC alone). Thus, all subjects in the study received the SOC, but some subjects received Multikine before the SOC (the two study treatment arms) and some did not (study control arm). In the third arm (3/7), which was study control arm, subjects received only the SOC (with no Multikine or CIZ). (CIZ comprised a non-chemotherapeutic dose of cyclophosphamide (administered one-time only IV-bolus, 3 days prior to the 1 st dose of Multikine), and indomethacin and zinc-multivitamins daily from day 1 of Multikine administration to one day before surgery to enhance Multikine activity.) In the second arm (1/7), subjects received the three-week Multikine regimen without CIZ prior to receiving the SOC.
#EARLY STAGE CANCER LYMPH NODES IN NECK PLUS#
In the primary treatment arm (3/7) subjects received three consecutive weeks of treatment with supraphysiologic doses of Multikine injected 5x/wk peritumorally and perilymphatically plus “CIZ” prior to receiving the SOC.
#EARLY STAGE CANCER LYMPH NODES IN NECK TRIAL#
The global IT-MATTERS trial was conducted in 23 countries in accordance with Good Clinical Practices, International Counsel for Harmonization standards, and all other country-specific regulatory requirements.įollowing diagnosis, subjects were randomized into one of three treatment arms. Multikine is the first investigational cancer immunotherapy being developed as a first-line neo-adjuvant treatment to be provided to previously untreated locally advanced primary disease SCCHN patients before they receive the SOC.
Multikine is a mixture of naturally occurring cytokines that regulate the immune system. The 928-patient IT-MATTERS study was designed to determine if Multikine provided survival and other clinical benefits to patients suffering from locally advanced primary squamous cell carcinoma of the head and neck (SCCHN), oral cavity and soft-palate.

) Additional results will be published in scientific journals and presented in scientific forums in the future. Some of the trial’s results were published in two peer-reviewed abstracts and a poster at the ASCO conference in June 2022. National Library of Medicine at the National Institutes of Health. is the largest clinical trial database in the world. VIENNA, Va.–( BUSINESS WIRE)–August 19, 2022–CEL-SCI Corporation (NYSE American: CVM) announced today that the results of its 10-year IT-MATTERS pivotal Phase 3 clinical trial in head and neck cancer with its investigational immunotherapy Multikine®* (Leukocyte Interleukin, Injection) have been posted on per U.S. CEL-SCI Corporation Announces Phase 3 Multikine® Head and Neck Cancer Results Posted on
